Turning Community Voices Into Regulatory Impact
Mobilizing the FSGS Community During a Critical FDA Review
When a critical moment arrived for the focal segmental glomerulosclerosis (FSGS) community, NephCure moved quickly to ensure the voices of patients, caregivers, clinicians, and advocates were heard.
Download the FSGS Community Petition to the FDAIntroduction
In January 2026, the U.S. Food and Drug Administration (FDA) was reviewing an application for sparsentan as a potential treatment for focal segmental glomerulosclerosis (FSGS), a rare and progressive kidney disease that can lead to kidney failure.
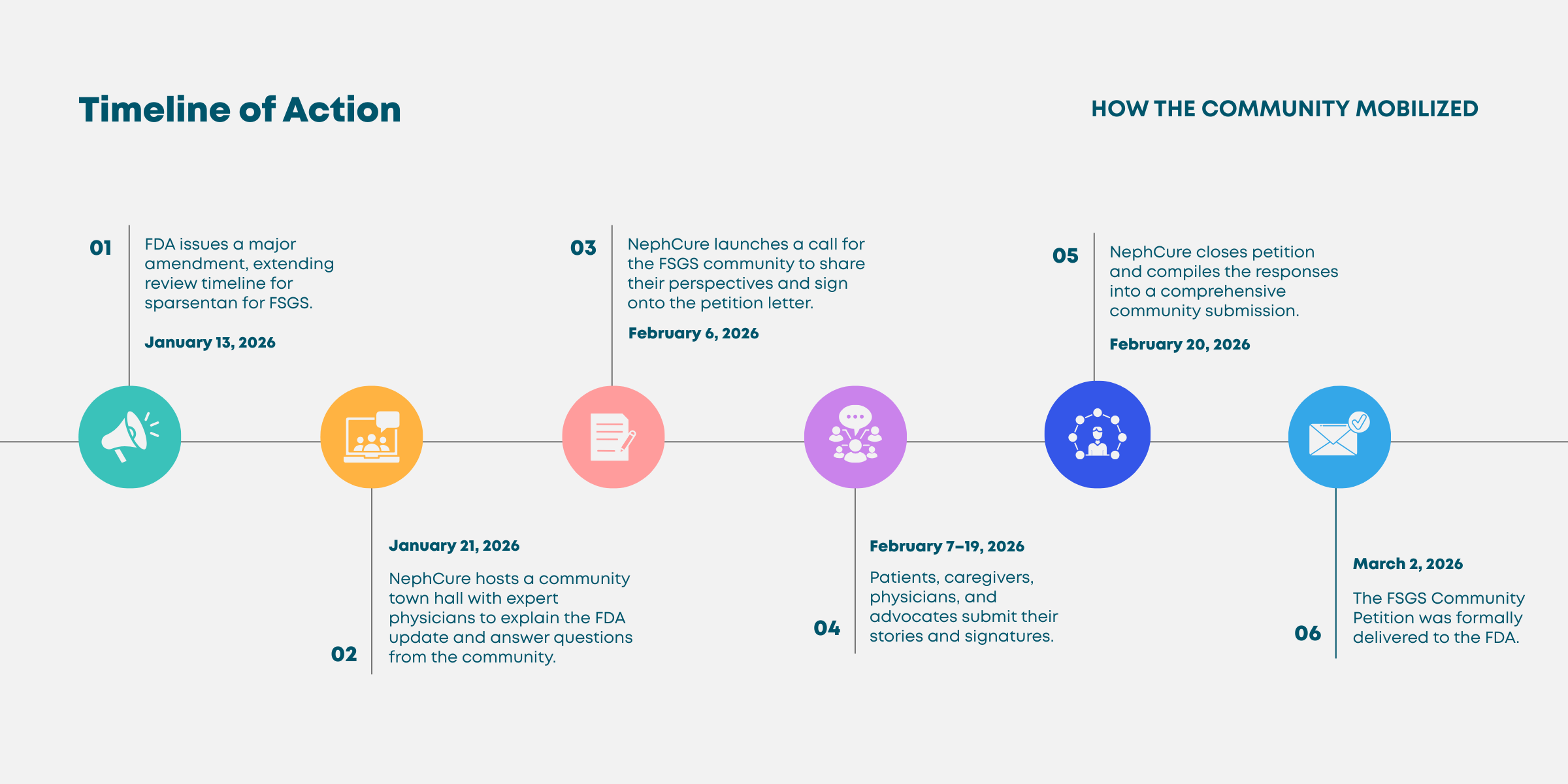
The FDA was expected to make a decision on January 13, 2026. On that date, they issued a major amendment, extending the review timeline to allow for more time to evaluate the data. The amendment movedthe FDA’s target decision date to April 13, 2026.
Moments like this are critical for the rare disease community. During regulatory reviews, the FDA evaluates clinical trial data, but it also considers the lived experiences of patients and the real-world impact of disease.
NephCure recognized this as an opportunity, and responsibility, to ensure the voices and experiences of the people living with FSGS were heard and part of the broader conversation.
Within just four weeks, NephCure launched a community petition, invited individuals across the FSGS, and rare kidney disease, community to share their perspectives on the urgent need for additionaltreatment options, and compiled those voices into a formal submission delivered to the FDA.
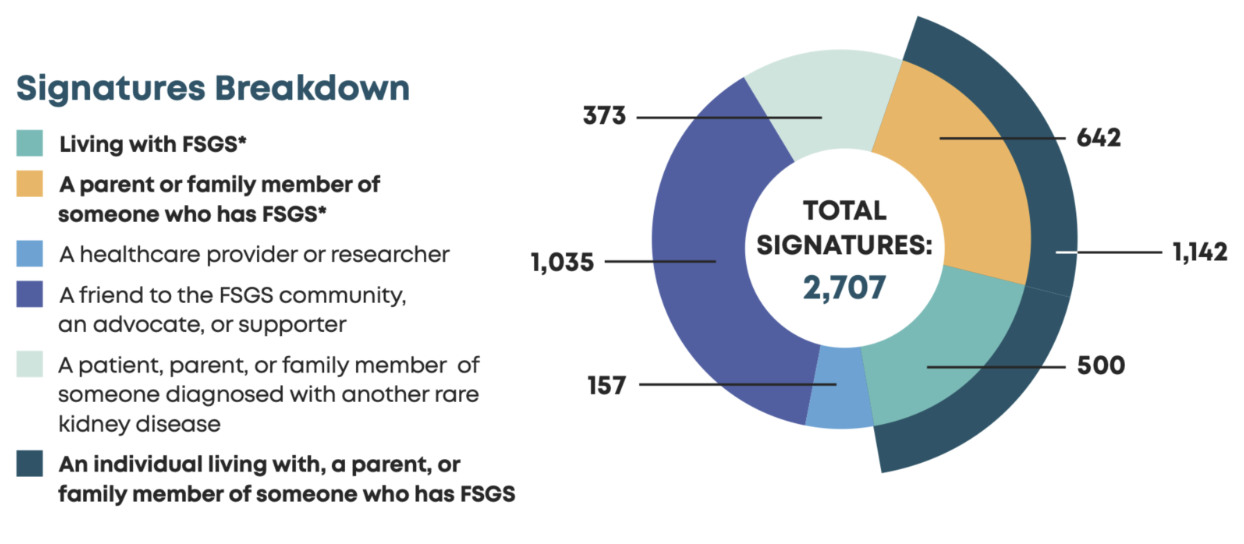
Community Response
2,700+
Signatures Submitted
1000+
Comments from the FSGS Community
125+
Comments from Healthcare Providers
1
Unified Message: The FSGS community urgently needs more treatment options

Read the FSGS Community Petition Submitted to the FDA
The packet NephCure submitted to the FDA reflects the voices, experiences, and hopes of individuals across the FSGS community.
By sharing their stories, community members helped ensure regulators could better understand the real-world impact of FSGS and urgent need for additional treatment options.
Download the Full Submission
This Effort was Powered by You
This submission would not have been possible without the people who stepped forward to share their personal experiences.
Patients, caregivers, clinicians, researchers, and advocates came together to ensure that the realities of living with FSGS were heard.
Your voices help drive progress for the entire rare kidney disease community.